Evaluation of antioxidant potential of methanolic extracts of bark and leaf of Quassia indica (Gaertn.) Nooteb
Keywords:
Quassia indica, Antioxidant, ABTS, DPPH, DPPHNO, Reducing power assay, Superoxide scavengingAbstract
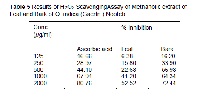
Plants are a natural source of secondary metabolites having antioxidant property and these compounds protect cells against adverse effects of reactive oxygen species. In the present study the methanolic extracts of bark and leaf of medicinal plant - Quassia indica (Gaertn.) Nooteb. were evaluated and compared employing ABTS cation decolorization test, DPPH scavenging assay, reducing power and scavenging potency towards hydrogen peroxide, nitric oxide and super oxide. The results suggested that the bark extract has better antioxidant efficacy than leaf for all in vitro models compared. The IC50 metrics (mg/g) of bark and leaf for the assays were ABTS (bark 552.36; leaf 1917); DPPH (bark 572.30; leaf 1782.31); H2O2 (bark 566.89; leaf 1904.32); Nitric Oxide (bark 956.75; leaf 1711.74) and Superoxide (bark 235; leaf 506.68) respectively. Results of the study concluded that Q. indica is a promosing source of antioxidants and could be exploited as a safe and stable natural agent for pharmaceutical preparations.
References
Viswanad VN. Aleykutty A. Zacharia SM, Thomas, L. Evaluation of Antioxidant and Free Radical Scavenging activity of Samadera indica using in vitro models. Phcog J,2011;3:85-90.
Raja S, Ravindranadh, K. Chemical and Pharmacological aspects of Samadera indica (Simaroubaceae): An Overview. Int J Biol Pharm Res,2014;5:958-963.
Chang CL, Lin CS, Lai, GH. Phytochemical characteristics, free radical scavenging activities, and neuroprotection of five medicinal plant extracts. J Evid Based Complemrntary Altern Med, Article ID 984295, 2012, 8 pages.
Masoko P, Eloff JN. Screening of twenty-four South African Combretum and six Terminalia species for antioxidant activites. Afr J Tradit Complement Altern Med,2007;4:231–239.
Hodzic Z. Pasalic H. Memisevic A, Srabovic M. Saletovic M, Poljakovic, M. The influence of total phenols content on antioxidant capacity in the whole grain extracts. Eur J Sci Res,2009;28:471–477.
Mittler R. Vanderauwera S, Gollery M, Van Breusegem F. Reactive oxygen network of plants. Trends Plant Sci,2004;9:490-498.
Re R, Pellegrini N, Proteggente A, Pannata A, Yang M, Rice-Evans C. Antioxidant activity applying an improved ABTS radical Decolorization Assay. Free Radic Biol Med,1999;26:1231-1237.
Blois MS. Antioxidant determination by the use of a stable free radical. Nature, 1958;29:1199 – 1200.
Ruch RJ. Cheng SJ, Klaunig JE. Prevention of Cytotoxicity and Inhibition of Intracellular Communication by Antioxidant Catechins isolated from Chinese green tea. Carcinigenesis,1989;10:1989, 1003.
Garrat DC. The Quantitative analysis of Drug, Chapman and Hall Ltd. Japan. Vol. 3, 1964:456-458.
Beauchamp C, Fridovich I. Superoxide dismutase: Improved assays and an assay applicable to Acrylamide gels. Anal Biochem,1971;44:276-277.
Oyaizu M. Studies on product of browning reaction: Antioxidative activities of products of browning reaction prepared from Glucose amine. Jpn J Nutr Diet,1986;44: 307-315.
Bijaya LM, Bikash B. Antioxidant capacity and Phenolics content of some Nepalese Medicinal Plants. Am J Plant Sci,2013;4:1660–1665.
Sumathy R, Sankaranarayanan S, Bama P. Ramachandran J, Vijayalekshmi M, Deevaraman M. Antioxidant and Antihemolytic activity of flavonoids- extract from fruit peel of Punica granatum. Asian J Pharmaceut Clin Res,2013; 6: 211-214.
Kokate CK. A Textbook for Practical Pharmacognosy, 2005 5th Ed.
Harborne JB. Phytochemical Methods, Chapman Hall, London, 1998; 60–66.
Nahak G, Sahu RK. In vitro antioxidative acitivity of Azadirachta indica and Melia azedarach Leaves by DPPH scavenging assay. Nat Sci,2010;8:23-28.
Alves CQ, David JM, David JP, Bahia MV, Anguilar RM. Methods for determination of in vitro antioxidant activity for extracts and organic compounds. Quim Nova,2010;33:2202-2210.