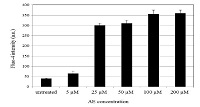
Aloe-emodin triggers ROS and Ca 2+ production and decreases the levels of mitochondrial membrane potential of human brain capillary endothelial cells
Keywords:
aloe-emodin, human brain capillary endothelial cells, reactive oxygen species, mitochondria, intracellular Ca 2Abstract
The aim of this work was to investigate the mechanisms of cytotoxicity of phyto-hydroxyanthraquinone aloe-emodin (AE) on human brain microvascular endothelial cell line hCMEC/D3 and to assess the cellular response in the early stage of treatment in order to extend the knowledge of AE’s anti-angiogenic properties. The immortalized human brain capillary endothelial cells hCMEC/D3 were treated with a series of AE concentrations (5 - 200 μM) for a period of 24 hours. The cell viability was determined by MTS assay. The cellular adenosine triphosphate (ATP) levels were evaluated by CellTiter-Glo® luminescent assay. The intracellular reactive oxygen species (ROS) were determined by 2’,7’-dichlorofluorescein (CM- H2DCFDA) fluorescence assay. The mitochondrial membrane potential (MMP) was assessed using tetramethylrhodamine methyl ester (TMRM) staining, while Fluo-4 was used to measure 2 the intracellular free Ca 2+ concentrations inside living cells analysed by High Content Analysis using the Arrayscan VTI 740. Twenty-four- hour treatment of hCMEC/D3 cells with AE, in concentrations between 50 and 200 µM, decreased the cell viability as well as the intracellular ATP levels in a dose- dependent manner. Increased ROS production and disruption of the mitochondrial membrane potential have also been detected. Notably, AE at a concentration greater than 5 µM dramatically increased intracellular calcium levels. Our results collectively indicate that AE inhibits proliferation of human brain microvascular cells via a mechanism involving ROS generation, disruption of Ca 2+ homeostasis and mitochondrial damage.
References
. Jain RK. Angiogenesis in brain tumours. Nat. Rev. Neurosci. 2007;8:610-22.
. Armstrong TS. Management of treatment-associated toxicites of anti-angiogenic therapy in patients with brain tumors. Neuro. Oncol. 2012;14:1203-14.
. Ng KW. Anti-angiogenic and cytotoxicity studies of some medicinal plants. Planta Med. 2010;76:935-40.
. Sagar SM. Natural health products that inhibit angiogenesis: a potential source for investigational new agents to treat cancer—Part 1. Curr. Oncol. 2006;13: 14–26.
. Zhou W. Pharmacological potential action of Rhein and its diverse signal transduction: A Systematic Review. World Journal of Pharmaceutical Research. 2014;3:3599-3626.
. Ribatti D. Endogenous inhibitors of angiogenesis: a historical review. Leuk Res. 2009;33: 638-44.
. He ZH. Anti-angiogenic effect and mechanism of rhein from Rhizoma Rhei. Phytomedicine. 2011;18:470-8.
. Rodriguez-Nieto S. Antiangiogenic activity of aeroplysinin-1, a brominated compound isolated from a marine sponge. FASEB J. 2002;16:261–263.
. Thomson RH. Naturally occurring quinones. Academic Press Inc. New York Book. 1971;1–38.
. Cárdenas C. Evaluation of the anti-angiogenic effect of aloe-emodin. Cell. Mol. Life. Sci. 2006;63:3083-9.
. Suboj P. See comment in PubMed Commons belowAloe emodin inhibits colon cancer cell migration/angiogenesis by down regulating MMP-2/9, Rho B and VEGF via reduced DNA binding activity of NF-κB. Eur. J. Pharm. Sci. 2012;45: 581-91.
. Chen R. See comment in PubMed Commons below Potential antineoplastic effects of Aloe-emodin: a comprehensive review. Am J Chin Med. 2014;42:275-88.
. Wang XH. Inhibitory effects of emodin on angiogenesis. Yao Xue Xue Bao. 2004; 39:254-8
. Lin SY. Emodin induces apoptosis of human tongue squamous cancer SCC-4 cells through reactive oxygen species and mitochondria-dependent pathways. Anticancer Res. 2009; 29:327-35.
. Amin IM. Involvement of Intracellular Calcium and MAPK in Aloe Emodin Induced Anti-Proliferative in MCF-7. ASM Science Journal. 2014;8:165–173.
. Weksler BB. Blood-brain barrier-specific properties of a human adult brain endothelial cell line. FASEB J. 2005;19:1872–1874.
. Wang P. Limitations of MTT and MTS-based assays for measurement of antiproliferative activity of green tea polyphenols. PLoS One. 2010;5:10202.
. Bexiga MG. Cationic nanoparticles induce caspase 3-7- and 9-mediated cytotoxicity in a human astrocytoma cell line. Nanotoxicology. 2011;5:557-67.
. Bagkos G. A new model for mitochondrial membrane potential production and storage. Med Hypotheses. 2014;83:175-81.
. Rasola A, Bernardi P. Reprint of “The mitochondrial permeability transition pore and its adaptive responses in tumor cells”. Cell Calcium. 2015;56:437–445.
. Camello-Almaraz C. Mitochondrial reactive oxygen species and Ca2+ signalling. Am. J. Physiol. Cell Physiol. 2006; 291:1082–1088.
. Görlach A. Calcium and ROS: A mutual interplay. Redox Biol. 2015;6:260-71.
. Huang FJ. Characterization of apoptosis induced by emodin and related regulatory mechanisms in human neuroblastoma cells. Int J Mol Sci. 2013;14:20139-56.
. Lyng FM. The involvement of calcium and MAP kinase signalling pathways in the production of radiation-induced bystander effects. Radiat Res. 2006; 165:400-9.