Mechanism of action of a bioassay-guided aqueous fraction of Pterocarpus marsupium Roxb hardwood on glucose-dependent insulin secretion
Keywords:
bioassay-guided, Pterocarpus, hardwoodAbstract
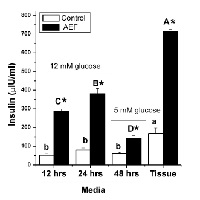
We previously demonstrated that a high molecular weight aqueous fraction of Pterocarpus marsupium Roxb. (PM) hardwood has insulinotrophic properties. However, the pharmacological mechanisms by which this fraction modulates insulin secretion remained unknown. The present study therefore isolated the insulinotrophic activity enriched fraction (AEF) from aqueous extract of PM and examined its pharmacological mechanisms. A bioassay method utilizing insulin secretion from mouse pancreas in vitro, was used to fractionate the insulinotrophic activity of PM and to delineate its pharmacological mechanisms. In addition, the effect of AEF on glucose clearance in normoglycemic, non-diabetic sheep in vivo was examined. The AEF mimicked the effect of sulphonylureas on insulin secretory pathways and modulated insulin biosynthesis. However, unlike tolbutamide, AEF-induced insulin secretion is glucose-dependent. Furthermore, three daily intravenous administrations of AEF had prolonged effects on glucose responsiveness in non-diabetic normal sheep. The use of AEF to combat the adverse effects of hyperglycemia appears to be beneficial by enhancing and sustaining the glucose-dependent insulin secretion processes in pancreas. Of note, the insulinotrophic effect of AEF is prolonged by many hours to days, unlike the numerous conventional insulin secretagogus which over stimulate the β-cells or pose a risk of hypoglycemia.
References
Guariguata L, Whiting DR, Hambleton I, Beagley J, Linnenkamp U, Shaw JE. Global estimates of diabetes prevalence for 2013 and projections for 2035. Diabetes Res Clin Pract 2014; 103(2): 137-149.
Kahn SE, Cooper ME, Del Prato S. Pathophysiology and treatment of type 2 diabetes: perspectives on the past, present, and future. Lancet 2014; 383(9922): 1068-1083.
Seino S, Shibasaki T, Minami K. Dynamics of insulin secretion and the clinical implications for obesity and diabetes. J Clin Invest 2011; 121(6): 2118-2125.
Grover JK, Yadav S, Vats V. Medicinal plants of India with anti-diabetic potential. J Ethnopharmacol 2002; 81(1): 81-100.
Modak M, Dixit P, Londhe J, Ghaskadbi S, Devasagayam TP. Indian herbs and herbal drugs used for the treatment of diabetes. J Clin Biochem Nutr 2007; 40(3): 163-173.
Rizvi SI, Mishra N. Traditional Indian medicines used for the management of diabetes mellitus. J Diabetes Res 2013; 2013: 712092.
Gupta SS. Effect of Gymnema sylvestre and Pterocarpus marsupium on glucose tolerance in albino rats. Indian J Med Sci 1963; 51: 716-724.
Chakravarthy BK, Gupta A, Gambhir SS, Gode KD. Pancreatic beta-cell regenaration. A novel antidiabtic mecanism of Pterocarpus marsupium Roxb. Indian J Pharmacol 1980; 12: 123-129.
Ahmad F, Khalid P, Khan MM, Chaubey M, Rastogi AK, Kidwai JR. Hypoglycemic activity of Pterocarpus marsupium wood. J Ethnopharmacol 1991a; 35(1): 71-75.
Grover JK, Vats V, Yadav S. Effect of feeding aqueous extract of Pterocarpus marsupium on glycogen content of tissues and the key enzymes of carbohydrate metabolism. Mol Cell Biochem 2002; 241(1-2): 53-59.
Dhanabal SP, Kokate CK, Ramanathan M, Kumar EP, Suresh B. Hypoglycaemic activity of Pterocarpus marsupium Roxb. Phytother Res 2006; 20(1): 4-8.
Halagappa K, Girish HN, Srinivasan BP. The study of aqueous extract of Pterocarpus marsupium Roxb. on cytokine TNF-alpha in type 2 diabetic rats. Indian J Pharmacol 2010; 42(6): 392-396.
Mohankumar SK, O'Shea T, McFarlane JR. Insulinotrophic and insulin-like effects of a high molecular weight aqueous extract of Pterocarpus marsupium Roxb. hardwood. J Ethnopharmacol 2012; 141(1): 72-79.
ICMR. Flexible dose open trial of Vijaysar in cases of newly-diagnosed non-insulin-dependent diabetes mellitus. Indian J Med Res 1998; 108(July): 24-29.
Chakravarthy BK, Gupta S, Gambhir SS, Gode KD. Pancreatic beta-cell regeneration in rats by (-)-epicatechin. Lancet 1981; 2(8249): 759-760.
Kolb H, Kiesel U, Greulich B, van der Bosch J. Lack of antidiabetic effect of (-)-epicatechin. Lancet 1982; 1(8284): 1303-1304.
Manickam M, Ramanathan M, Jahromi MA, Chansouria JP, Ray AB. Antihyperglycemic activity of phenolics from Pterocarpus marsupium. J Nat Prod 1997; 60(6): 609-610.
Anandharajan R, Pathmanathan K, Shankernarayanan NP, Vishwakarma RA, Balakrishnan A. Upregulation of Glut-4 and PPAR gamma by an isoflavone from Pterocarpus marsupium on L6 myotubes: a possible mechanism of action. J Ethnopharmacol 2005; 97(2): 253-260.
Kosaraju J, Dubala A, Chinni S, Khatwal RB, Satish Kumar MN, Basavan D. A molecular connection of Pterocarpus marsupium, Eugenia jambolana and Gymnema sylvestre with dipeptidyl peptidase-4 in the treatment of diabetes. Pharm Biol 2014; 52(2): 268-271.
Mohankumar S, McFarlane JR. An aqueous extract of Curcuma longa (turmeric) rhizomes stimulates insulin release and mimics insulin action on tissues involved in glucose homeostasis in vitro. Phytother Res 2011; 25(3): 396-401.
Gatford KL, De Blasio MJ, Thavaneswaran P, Robinson JS, McMillen IC, Owens JA. Postnatal ontogeny of glucose homeostasis and insulin action in sheep. Am J Physiol Endocrinol Metab 2004; 286(6): E1050-1059.
Rorsman P, Braun M. Regulation of insulin secretion in human pancreatic islets. Annu Rev Physiol 2013; 75: 155-179.
Newsholme P, Gaudel C, McClenaghan NH. Nutrient regulation of insulin secretion and beta-cell functional integrity. Adv Exp Med Biol 2010; 654: 91-114.
Henquin JC. Pathways in [beta]-cell stimulus-secretion coupling as targets for therapeutic insulin secretagogues. (Section 11: [beta]-cell therapeutic targets other than ATP- sensitive [K.sup.+] channels). Diabetes 2004; 53(12): S48.
Proks P, Reimann F, Green N, Gribble FM, Ashcroft FM. Sulphonylurea stimulation of insulin secretion. Diabetes 2002; 51(Suppl.3): S368-S376.
Trube G, Rorsman P, Ohno-Shozaku T. Opposite effects of tolbutamide and diazoxide on the ATP-sensitive potassium channel. . Lancet 1986; 2: 493–499.
Hermans MP, Schmeer W, Henquin JC. The permissive effect of glucose, tolbutamide and high (K.sup.+) on arginine stimulation of insulin release in isolated mouse islets. Diabetologia 1987; 30: 659-665.
Smith PA, Sakura H, Coles B, Gummerson N, Proks P, Ashcroft FM. Electrogenic arginine transport mediates stimulus-secretion coupling in mouse pancreatic beta-cells. J Physiol 1997; 499 ( Pt 3): 625-635.
Newsholme P, Brennan L, Rubi B, Maechler P. New insights into amino acid metabolism, beta-cell function and diabetes. Clin Sci (Lond) 2005; 108(3): 185-194.
Zammitt NN, Frier BM. Hypoglycemia in type 2 diabetes: pathophysiology, frequency, and effects of different treatment modalities. Diabetes Care 2005; 28(12): 2948-2961.
Doyle ME, Egan JM. Pharmacological agents that directly modulate insulin secretion. Pharmacol Rev 2003; 55: 105-131.
Bergman RN. Lily lecture 1989.Toward physiological understanding of glucose tolerance: Minimal model approach. Diabetes 1989; 38: 1512-1527.
Bracho-Romero E, Reaven GM. Effect of age and weight on plasma glucose and insulin responses in rat. J Am Geriatr Soc 1977; 25: 299-302.
Amiel SA, Sherwin RS, Simonson DC, Lauritano AA, Tamborlane WW. A contributing factor in poor glycemic control in adolescents with diabetes. New Eng J Med 1986; 315: 215-219.
Bloch CA, Clemons P, Sperling MA. Puberty decreases insulin sensitivity. J Pediatr 1987; 110: 481-487.