Chemical composition, antioxidant and antibacterial activities of extracts obtained from the roots bark of Arbutus andrachne L. a Lebanese tree.
Keywords:
Arbutus andrachne L., Ericaceae, roots bark extracts, antioxidant activity, ntibacterial activityAbstract
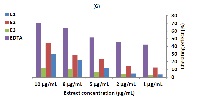
Context and purpose of the study: The leaves, fruits, barks and roots of Arbutus andrachne L (A. andrachne), have been adopted to have high therapeutic value resulting from the presence of antioxidant compounds such as flavonoids, phenolic and tannins. In the present work, three extracts obtained from A. andrachne roots bark were evaluated for their antioxidant and antibacterial activities .The total phenolic content, flavonoid, condensed tannins and anthocyanins were determined in order to correlate them with the antioxidant activity of extracts. Main findings: The highest amounts of phenolic and tannins were found in the ethyl-acetate, while the anthocyanins ones were highly observed in the methanol-water extract. The lowest IC50 values for DPPH (0.6 µg/mL), and metal chelating assay (13.45µg/mL) were recorded in the ethyl-acetate extract and the methanolic one respetively. Gram positive bacteria (S. aureus and E. faecalis) were more susceptible to the antimicrobial potential of the methanol extract, while E.coli and P. aeruginosa as Gram negative bacteria turned out to be more resistant to the same extract. The ethyl-acetate extract was more effective on E. faecalis than on S. aureus; while E. coli and P. aeruginosa were the most resistant to this extract. Brief summary and potential implications: An appropriate dose of antioxidants derived from A. andrachne bark of the roots extracts in the human diet can help to avoid the risk of contracting diseases where ROS are involved in the pathogenesis. In fact, phenolic compounds in these extracts are among the natural antioxidants being studied by the scientific community due to their biological properties, e.g., antioxidant and antimicrobial activities.
References
. Funda Sibel P, and Hakan G. The role of free radicals in ethiopathogenesis of diseases. Advances in Molecular Biology. 2008; 1:1-9.
. Lambeth JD. Nox Enzymes, ROS, and Chronic Disease: An Example of Antagonistic Pleiotropy. Free Radic Biol Med. 2007; 43(3): 332–347.
. Masella R, Di Benedetto R, Vari R. Novel mechanisms of natural antioxidant compounds in biological systems: involvement of glutathione and glutathione-related enzymes. J Nutr Biochem. 2005; 16: 577–586.
. Gülçin İ. Antioxidant activity of food constituents: an overview. Arch Toxicol. 2012; 86(3): 345-391.
. Nagendran B, Kalyana S, Samman S. Phenolic compounds in plants and agri-industrialby-products: Antioxidant activity, occurrence, and potential uses. Food Chemistry. 2006; 99: 191–203.
. Proestos C, Chorianopoulos N, Nychas G.-JE, Komaitis, M. RP-HPLC analysis of the phenolic compounds of plant extracts. Investigation of their antioxidant capacity and antimicrobial activity. J. Agric. Food Chem. 2005; 53: 1190–1195.
. Garcia-Lafuente A, Guillamon E. Villares A, Rostagno MA, Martinez JA. Flavonoids as anti-inflammatory agents: implications in cancer and cardiovascular disease. Inflamm. Res. 2009; 58: 537-552.
. Mostafa SE, Karam NS, Shibli RA, Alali FQ. Micropropagation and production of arbutin in oriental strawberry tree (Arbutus andrachne L.). Plant Cell Tiss Cult. 2010; 103: 111-121.
. Seker M. Constituents of Arbutus andrachne. Fitoterapia.1991; 62: 176-177.
. Şeker M and Toplu C. Determination and Comparison of Chemical Characteristics of Arbutus unedo L. and Arbutus andrachnae L. (Family Ericaceae) Fruits. Journal of Medicinal Food. 2010; 13(4): 1013-1018.
. Tawaha K, Alali F, Gharaibeh M., Mohammad M, El-Elimat T. Antioxidant activity and total phenolic content of selected Jordanian species. Food Chem. 2007; 104: 1372-1378.
. Singleton VL, Rossi JR. Colorimetric of total phenolics with phosphomolybdic–phosphotungstic acid reagents. Am. J. Enol. Viticult. 1965; 16: 144–158.
. Kim D, Chun O, Kim Y, Moon H, Lee C. Quantification of phenolics and their antioxidant capacity in fresh plums. J. Agric.Food Chem. 2003; 5: 6509-6515.
. Sun B, Ricardo-da-Silva J, Spranger I. Critical factors of vanillin assay for Catechins and Proanthocyanidins. J. Agric. Food Chem. 1998; 46: 4267-4274.
. Wolfe K, Wu X, Liu RH. Antioxidant activity of apple peels. J.Agric.Food Chem. 2003; 51: 609-614.
. Berker K, Guc lu K, Tor I, Apak R. Comparative evaluation of Fe (III) reducing power-based antioxidant capacity assays in the presence of phenanthroline, batho-phenanthroline, tripyridyltriazine (FRAP) and ferricyanide reagents. Talanta. 2007; 72: 1157–1165.
. Foti MC, Daquino C, Geraci C. Electron-transfer reaction of cinnamic acids and their methyl esters with the DPPH radical in alcoholic solutions. J.Org.Chem. 2004; 69: 2309-2314.
. Decker EA, Welch B. Role of ferritin as a lipid oxidation catalyst in muscle food. J. Agric. Food Chem.1990; 38(3): 674–677.
. Hammer, K.A., Carson, C.F. and Riley, T.V. In vitro activity of Melaleuca alternifolia (tea tree) oil against dermatophytes and other filamentous fungi. Journal of Antimicrobial Chemotherapy.2002; 50: 195–199.
. Edziri H, Ammar S, Souad L, Mahjoub MA, Mastouri M, Aouni M et al. In vitro evaluation of antimicrobial and antioxidant activities of some Tunisian vegetables. South Afr J Bot. 2012; 78: 252-6.
. Goli, AH, Barzegar M, Sahari MA. Antioxidants activity and total phenolic compounds of pistachio(Pistachia vera) hull extracts. Food Chem.2004; 92: 521-525.
. Abdille MH, Singh RP, Jayaprakasha GK, Jena BS. Antioxidant activity of the extracts from Dillenia indica fruits. Food Chemistry. 2005; 90(4): 891-896.
. Djabou N, Dib MEA, Allali H, Benderb A, Kamal AA, Ghalem S, Tabti B. Evaluation of antioxidant and antimicrobial activities of the phenolic composition of Algerian Arbutus unedo L. roots. Pharmacognosy J.2013; 5: 275-280.
. Orak HH, Yagar H, Isbilir SS, Demirci AS, Gumus T, Ekinci N. Evaluation of antioxidant and antimicrobial potential of strawberry tree (Arbutus Unedo L.)leaf. Food Science and Biotechnology.2011; 20(5): 1249-1256.
. Chavan UD, Shahidi F, Naczk M. Extraction of condensed tannins from beach pea (Lathyrus maritimus L.) as affected by different solvents. Food Chemistry. 2001; 75(4): 509-512.
. Kim MY, Seguin P, Ahn JK, Kim JJ, Chun SC, Kim EH, Seo SH, Kang EY, Kim SL, Park YJ, Ro HM, Chung IM. Phenolic compound concentration and antioxidant activities of edible and medicinal mushrooms from Korea. J. Agric. Food Chem.2008; 56: 7265–7270.
. Oliveira I, Sousa AS, Ferreira ICFR, Bento A, Estevinho L, Pereira JA. Total phenols, antioxidant potential and antimicrobial activity of walnut (Juglans regia L.) green husks. Food Chem. Toxicol.2008; 46: 2326–2331.
. Malheiro R, Sá O, Pereira E, Aguiar C, Baptista P, Pereira JA. Arbutus unedo L. leaves as source of phytochemicals with bioactive properties. Ind.Crops Prod. 2012; 37 (1): 473-478.
. Chun OK, Kim DO, Lee CY. Superoxide radical scavenging activity of the major polyphenols in fresh plums. J. Agric. Food Chem. 2003: 51; 8067–8072.
. De Graft-Johnson J, Kolodziejczyk K, Krol M, Nowak P, Krol B, Nowak D. Ferric Reducing Ability Power of Selected Plant Polyphenols and their Metabolites: Implications for Clinical Studies on the Antioxidant Effects of Fruits and Vegetable Consumption. Basic & Clinical pharmacology & Toxicology. 2007; 100(5): 345-352.
. Dib MA, Djabou, N, Allali H, Tabti B. Identification of phenolic compounds and antimicrobial activity of roots of Arbutus unedo L. Asian J.Chem. 2010; 22(5): 4045-4053.
. Afjal HM, Jayshree D, Hemanta KG, Pronobesh C, Satya BP. (2014). Antimicrobial activity of methanol extract and fractions from Sarcochlamys pulcherrima. Bangladesh J Pharmacol.2014; 9: 4-9.